PuraStat
Providing Clear Control
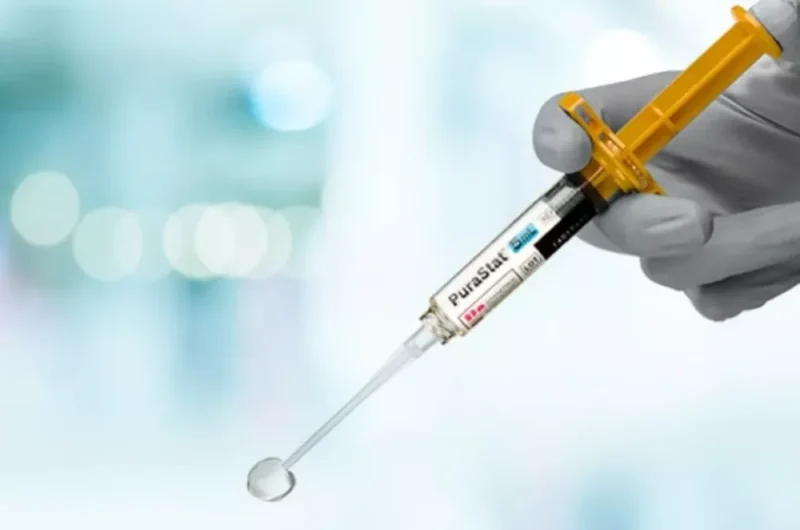
PuraStat is a synthetic haemostatic material in the form of a prefilled syringe.
PuraStat is indicated for haemostasis in the following situations encountered during surgery or endoscopic procedure, when haemostasis by ligation or standard means is insufficient or impractical1:
- Bleeding from small blood vessels and oozing from capillaries of the parenchyma and surrounding tissues of solid organs
- Oozing from vascular anastomoses to native or artificial vessels, on the surface of blood vessels and surrounding tissues
- Bleeding from small vessels and oozing from capillaries of the GI tract and surrounding tissues
- Reduction of delayed bleeding following gastrointestinal endoscopic submucosal dissection (ESD) procedures in the colon and rectum
-
Mimic Human ECM
-
Does Not Swell
-
No Assembly or Preparation
-
Revolutionary Bleeding Control
-
Resorbed in ~30 Days
-
Synthetic
-
Suturable
-
Transparent
-
Will Not Migrate
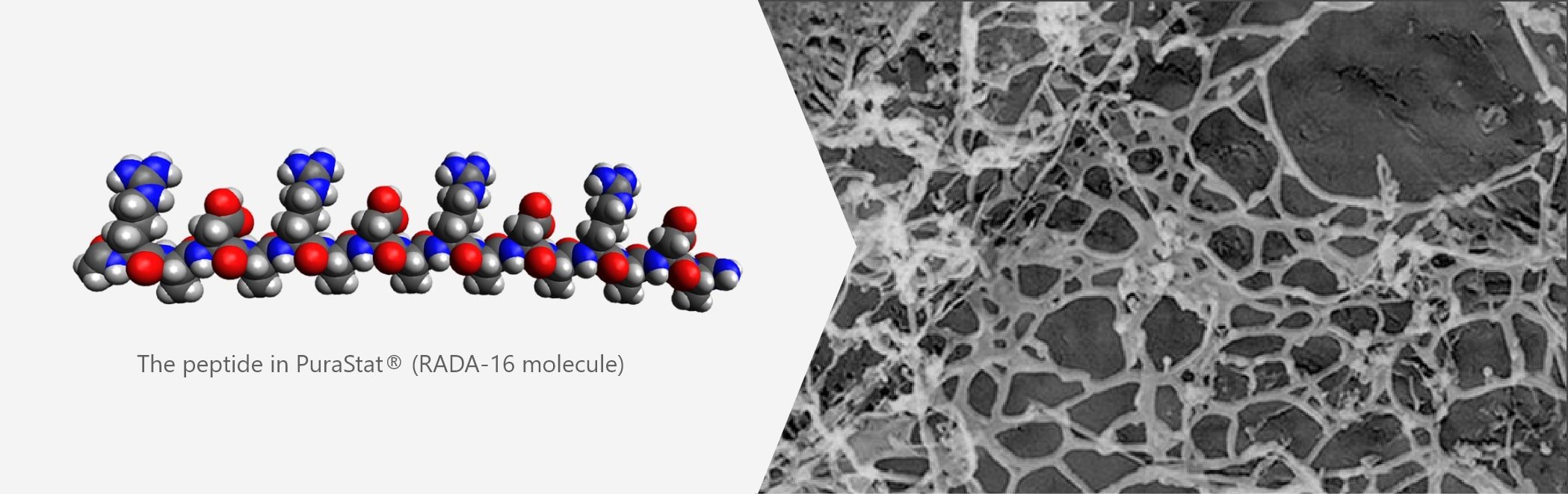
PuraStat mimics extra cellular matrix
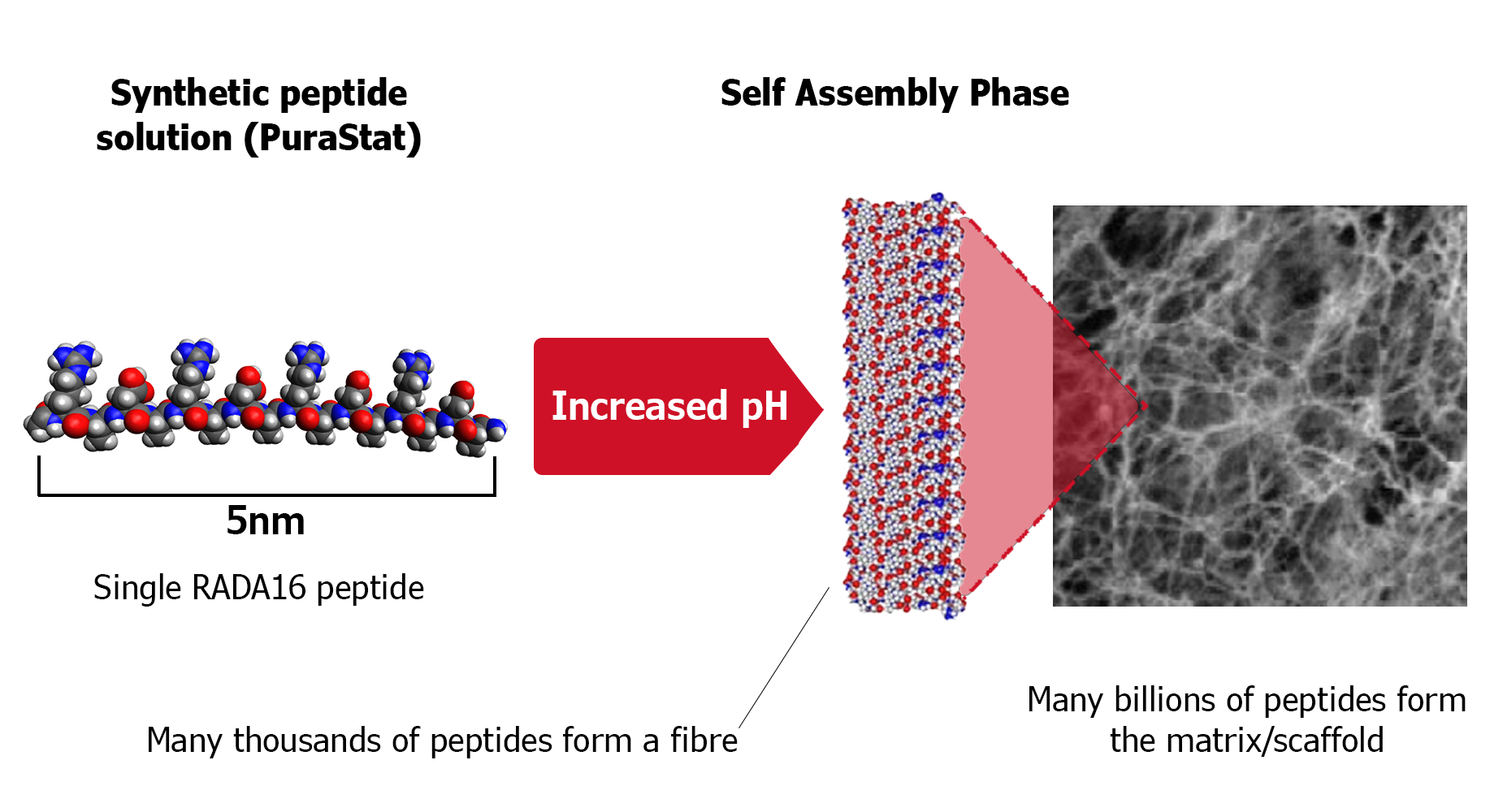
The peptides in PuraStat are synthetized in a specific sequence which provide the molecule with shape memory to enable them to self assemble into a scaffold which mimics human extra cellular matrix (ECM).
Based on results of scanning electron microscopy, the structure of PuraStat self-assembling peptide hydrogel is understood to be a network of fibres with a diameter of around 10–20 nm and pores with a diameter of about 50–200 nm that resembles the mesh structure of natural collagen.
- Dhwani J. , Rajesh V. A Review on Extracellular Matrix Mimicking Strategies for an Artificial Stem Cell Niche. Polymer Reviews. 2015, 55:4, 561-595, DOI:10.1080/15583724.2015.1040552
PuraStat does not swell1
In internal testing PuraStat does not swell after implantation or during the resorption process. This is because unlike other hydrogels which are designed to absorb water as part of their breakdown cycle, PuraStat is broken down by enzymatic action.
As PuraStat doesn’t swell it will not cause compression of underlying tissues of structures, or prevent that natural physiologic dilatation of vessels.
1. Data on file (Swell Report 2018, Eun Seok Gil 12/06/2018)
2. PuraStat Instructions for Use IFU-007 / IFU-011 Rev 2
PuraStat = No Assembly or Preparation
PuraStat is delivered in a prefilled syringe, just connect the applicator using the luer and the product is ready to use.
PuraStat has been demonstrated to achieve complete haemostasis in less than 15 seconds.1
1. Morshuis M, Final Clinical Study Report: A Single-Centre, Single Arm Post-Market Clinical Follow-Up to Confirm the Safety and Performance of PuraStat Absorbable Haemostatic Material for the Management of Bleeding After Left Ventricular Assist Device (LVAD) Implantation
Revolutionary Bleeding Control
The trigger for the peptides in PuraStat to begin self assembly is a change in ionic charge. PuraStat is acidic so when in contact with more alkaline liquids the reduction in ionic charge causes the rapid self assembly of β-sheets. The β-sheets then transition to form a complex network of fibres with a diameter of around 10–20 nm and pores with a diameter of about 50–200nm that resembles the mesh structure of natural collagen1. The formation of this complex fibre network rapidly seals the bleeding point leading to haemostasis.
1. Masuhara H. et al. Novel Infectious Agent-Free Hemostatic Material (TDM-621) in Cardiovascular Surgery Ann Thorac Cardiovasc Surg. 2012;18(5):444-51
PuraStat is Resorbed in 30 Days1
PuraStat is broken down by the actions of enzymes which break down the peptide scaffold into the constituent amino acids over approximately 30 days.1 The amino acids are metabolised by the body or excreted if they are surplus to requirements.
Breakdown Component
| Arginine Arg |
 |
A conditionally essential amino acid, it plays an important role in cell division, the healing of wounds, removing ammonia from the body, immune funciton, and the replease of hormones |
| Alanine Ala |
 |
A nonessential amino acid, meaning it can be manuratured by the human body, hence need not be obtained directly through diet. Alanine is found in a wide variety of foods |
| Asparatic Acid Asp |
 |
An acidic amino acid that is a constituent of most proteins and also occurs in sugar cane. It is important in the metabolism of nitrogen in animals and also acts as a neurotransmitter |
1. PuraStat IFU-007 Rev 2 / IFU-011 Rev 2
PuraStat is 100% Synthetic
PuraStat contains no glutaraldehyde.
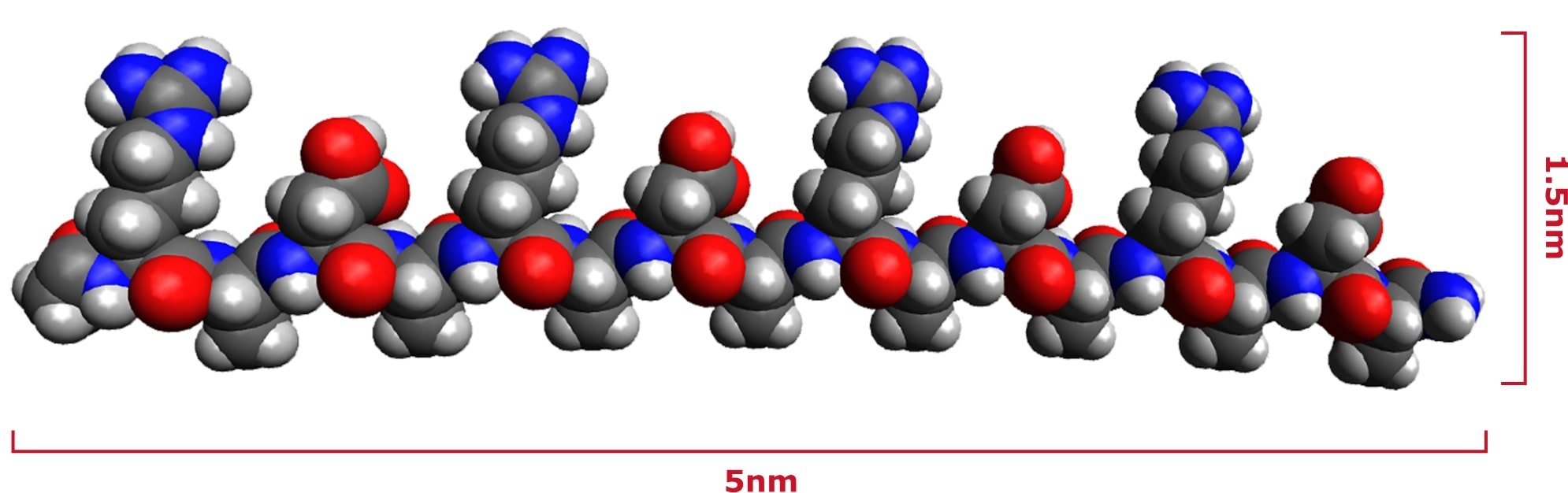
The peptides are made from chemically synthesized amino acids to guarantee the final product is contamination free. The amino acids are bonded together in a specific sequence at a 2.5% concentration in aqueous solution to make PuraStat.1
1. PuraStat IFU-007 Rev 2 / IFU-011 Rev 2
PuraStat is Suturable & Clipable
Because PuraStat is a liquid hydrogel, it is largely composed of water and remains completely flexible once applied. Because it does not harden or cure it remains soft and can easily be sutured through should the need arise.
This also means that you can place surgical or endoscopic clips directly through a layer of PuraStat, this is very helpful if using PuraStat to clearly identify bleeding points when bleeding is more profuse or persistent.
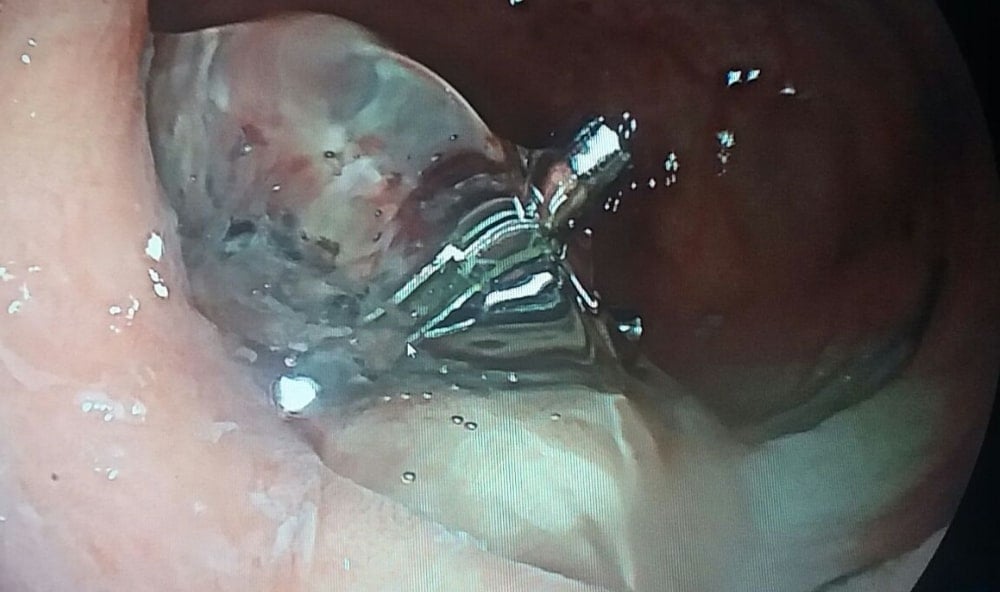
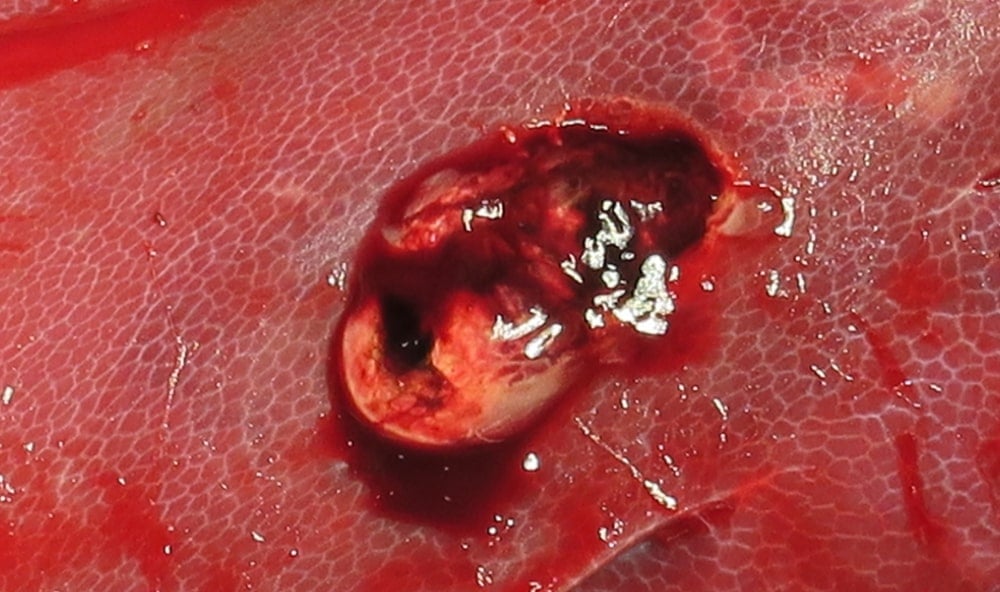
PuraStat is Transparent
This image demonstrates the transparency of PuraStat once applied clear visualisation of the underlying tissue is maintained.
PuraStat Will Not Migrate1
PuraStat is naturally viscous. When in contact with blood the change in pH causes the self assembly of PuraStat> to occur rapidly at the interface between it and the bleeding point, gaining further integral strength as the reaction occurs. Any PuraStat not incorporated in the final clot will separate and may be left in situ.

PuraStat Biologic Safety Report – Current Version
PuraStat Technology
The matrix is formed from synthetic peptides which are composed of three naturally occurring amino acids in a sequence which is repeated 4 times. The peptides self-assemble to form a 3 dimensional matrix upon a change in pH2 caused by exposure to an ionic solution such as blood, peritoneal fluid or other bodily secretions.
Applying PuraStat is simple
PuraStat is a haemostat existing of a nanofiber network structure that requires no preparation. This means it is ready to apply straight from packet to patient.
To apply PuraStat is just as easy, simply deploy PuraStat at the base of the lesion and wait, following the instructions below:
- Remove as much blood as possible from haemorrhagic site.
- Apply an adequate quantity of PuraStat to the haemorrhagic site.
- The nozzle should be as close as possible to the bleeding point.
- Do not disturb the self-assembled PuraStat gel until sufficient time has been allowed for haemostasis to occur.
- If necessary, repeat the application of PuraStat several times until haemostasis is achieved.
- After haemostasis is confirmed, excess PuraStat may be left in place.
A global network of distributors
We have a global network of partners and distributors who work closely with us to ensure you safely get our products and full, high quality support wherever you are and whenever you need it. They all believe in our products as much as we do, and can’t wait to show you how they can help you

Keep up to date with PuraStat

.png?width=73&height=73&name=calendar%20(1).png)
Reference
1. PuraStat IFU-007 Rev 2 , PuraStat IFU-011 Rev 2, PuraStat IFU-018 Rev 1
Important Note: The current IFU outside of Europe depends on the registration status. The approved indication can differ in your country, please contact 3-D Matrix if you need any additional information on device indication.
Products from 3-D Matrix
Accessories
Our accessories are specifically developed with users in mind for both endoscopic and laparoscopic procedures, giving you the full set of accessories that you need for your procedures.
Simply connect the PuraStat syringe with the accessory via the leur lock connector** and it is ready to use.
If you are interested in our accessories or would like more information, please contact us.

PuraStat
PuraStat is a synthetic haemostatic material in the form of a prefilled syringe. PuraStat is indicated for haemostasis in the following situations encountered during surgery, when haemostasis by ligation or standard means is insufficient or impractica1:
Current Page
PuraBond
PuraBond is a synthetic haemostatic material in the form of a prefilled syringe, filled with a clear, aqueous peptide solution. PuraBond is part of the RADA16 family of products, alongside PuraStat. Contact between the product and liquids such as blood leads to the formation of fibres in the aqueous solution, yielding a peptide hydrogel. The hydrogel quickly coats the point of bleeding and, by physically closing the superficial part of the broken blood vessel, causes blood coagulation in the deeper part of the vascular wall, resulting in haemostasis.
Learn More »
PuraShield
PuraShield is a synthetic haemostatic material for ENT surgery. A ready-to-use, slightly viscous hydrogel, composed of self-assembling peptides (SAPs) founded by the Massachusetts Institue of Technology (MIT), whilst undergoing tissue regeneration research. PuraShield is licenced in Spain as a medical device for use in Ears, Nose, Throat, Head and Neck surgeries.
PuraShield has many advantages which lend it to being a suitable bleeding management solution for FESS, adult tonsillectomies, thyroidectomies and amongst many other types of surgery. PuraShield's composition mimics the human extracellular matrix, and it does not cause swelling of the tissue. Its transparency and viscosity ensure precise control while maintaining visibility during the surgery, even in the event of working in small and narrow spaces.

